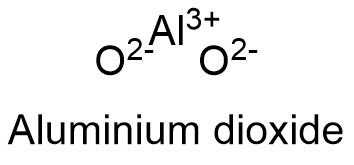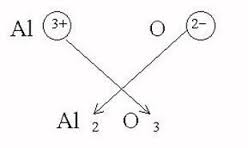
What is the formula for aluminum oxide? What is the balanced equation for the formation of aluminum oxide? | Socratic

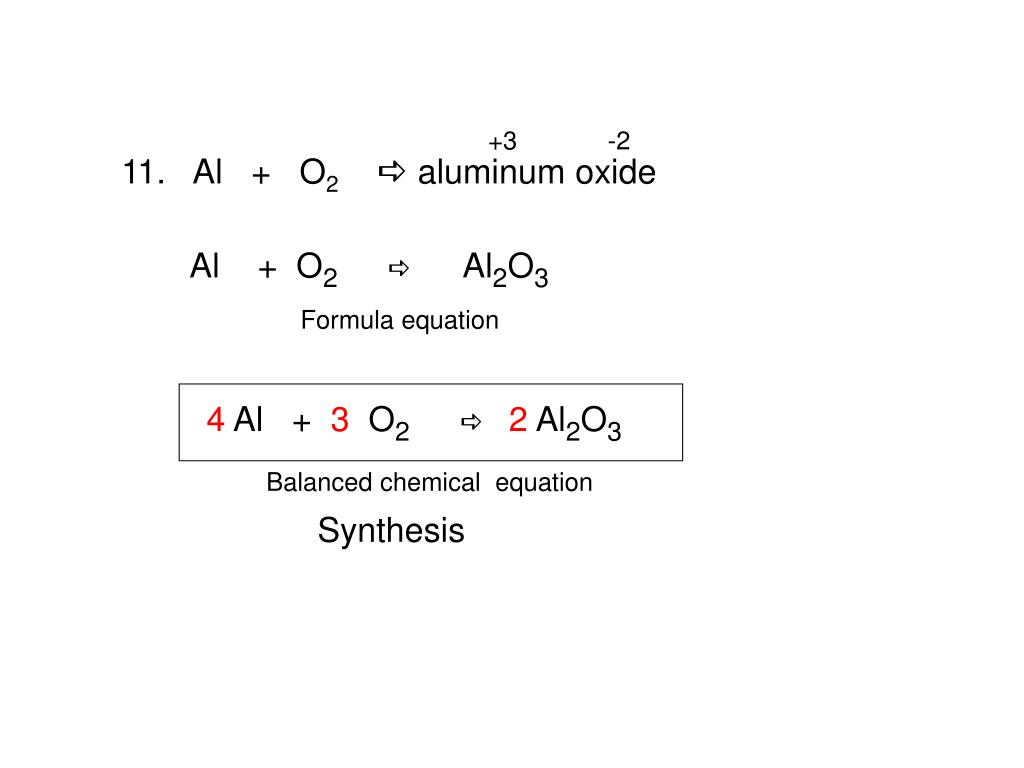
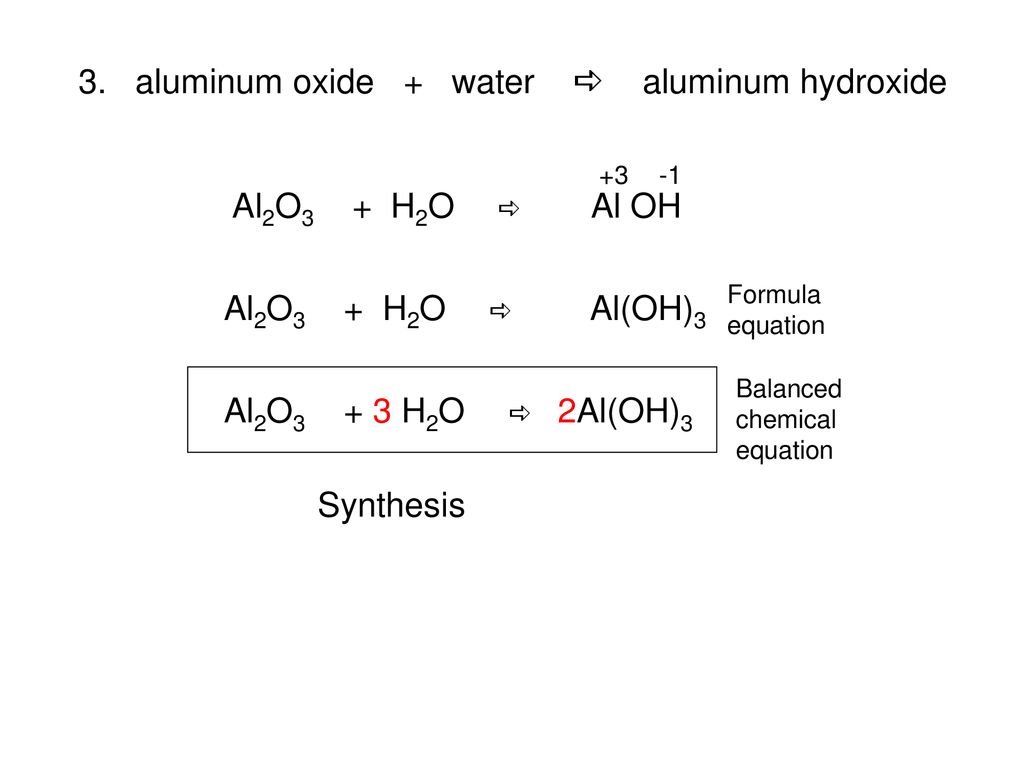
Practice Write a balanced formula equation for: aluminum + oxygen aluminum oxide Al +Al OO2 →O2 →Al 2 O ppt download

Please Help!) Aluminum reacts with oxygen to produce aluminum oxide (AI203). The balanced chemical - brainly.com

Solid manganese(IV) oxide reacts with the solid aluminum metal to produce solid manganese and solid - brainly.com

PPT - Writing Chemical Equations to Symbolize Chemical Reactions PowerPoint Presentation - ID:4566911



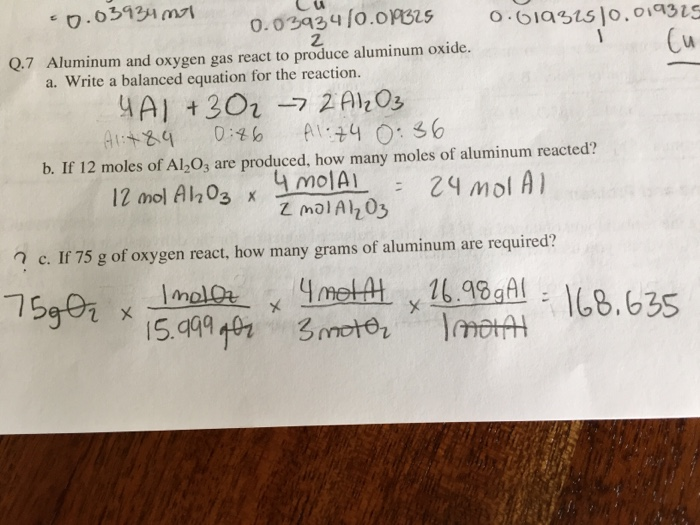






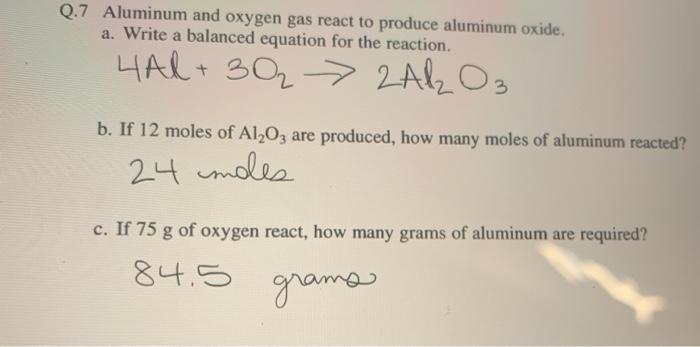
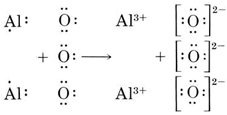
![Kannada] Write chemical equations that show aluminium oxide reacts wi Kannada] Write chemical equations that show aluminium oxide reacts wi](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/7529731.webp)